In this project our group had to design a board game using chemical reactions. We were given a board and our directions were to use chemicals we had used in our outside of class to use in the board game. We chose to do a winter wonderland theme because one of our reactions created a snow like substance. We had decorations including presents, 3-D printed game pieces, and much more! Below are our pictures, solutions, and terms that we needed to know or use. The formulas for reactions we used are in the key terms section.
These are our game rules for our board game. Listed inside are rules and safety cautions for our players.
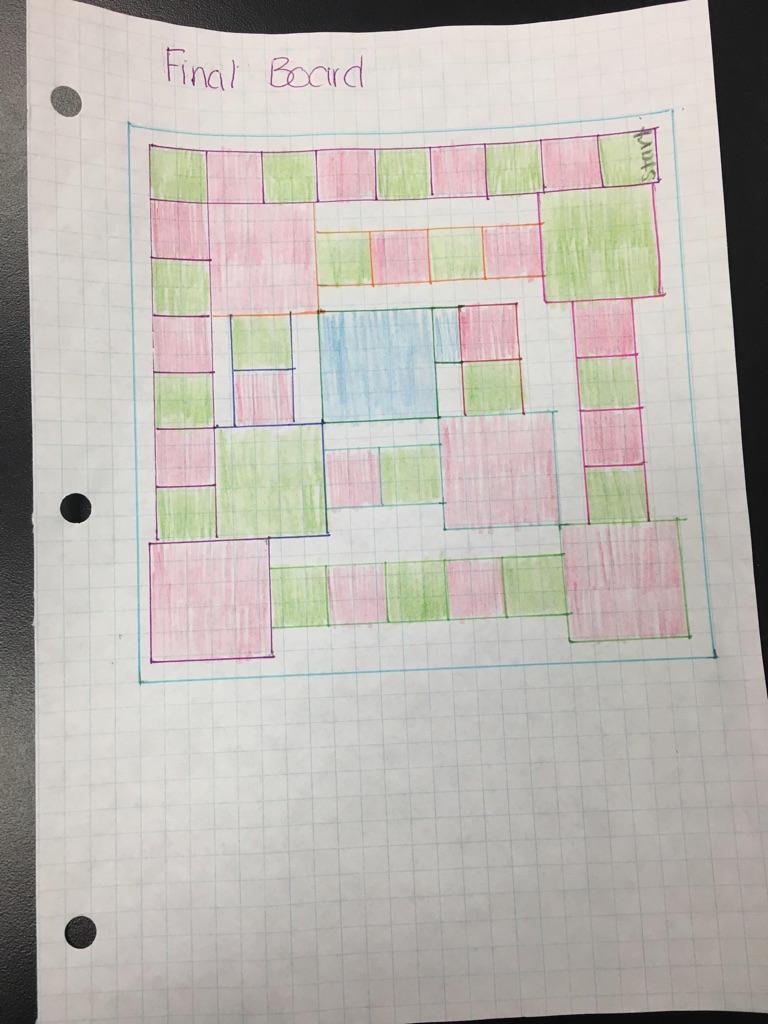
This is a picture of our whole board game. We had six reaction with one duplicate making seven stations. We had a start and the finish line was the center piece with the tree. We decided to start at the bottom and end at the top to signify the spiral from the bottom of a tree to the top.
Here are our safety precautions
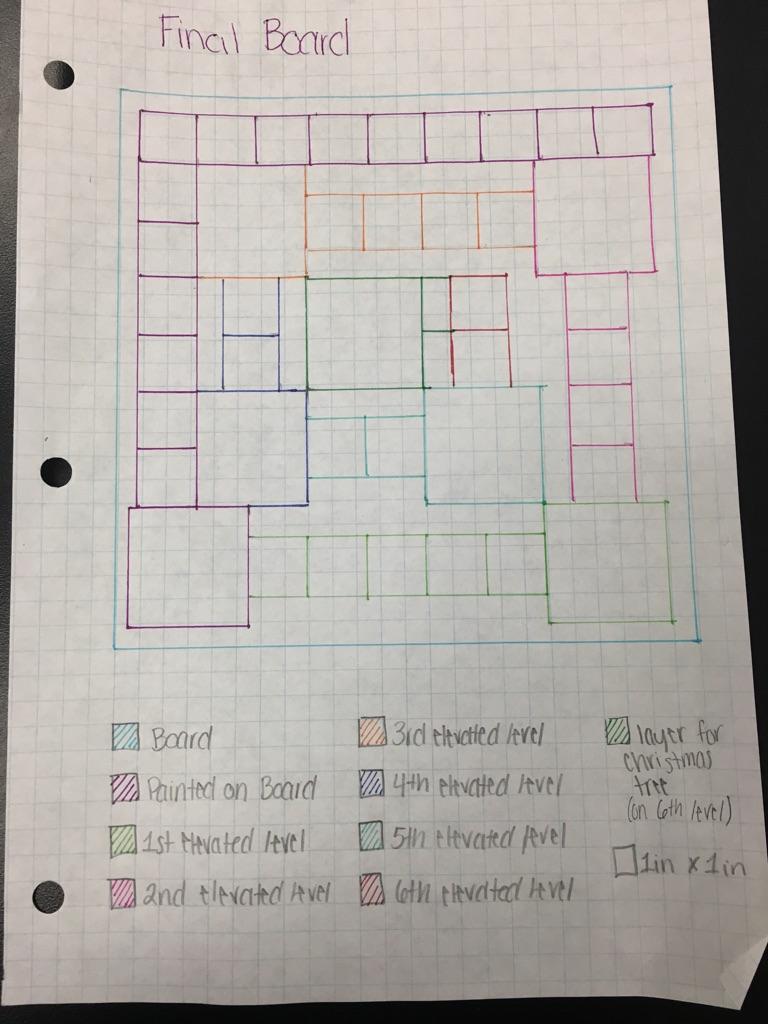
Our board schematics.
Reaction 1:The two petri dishes in the top left of the picture are our present reaction. We used Copper Chloride with aluminum wrapped presents. This is a single replacement reaction. The Aluminum broke up, revealing an eraser which we dipped in water and gave to the players.
Reaction 2 and 6: This reaction is our snow part. We mixed polyacrylic acid with water to create our snow. This was a physical reaction because the polymers expanded from the water, which can be reversed when dried out.
Reaction 3: This reaction was a mixture of baking soda (sodium bicarbonate) and vinegar (acetic acid), which let off carbon dioxide to inflate the balloon. This was our production of a gas.
Reaction 4: The copper chloride and aluminum react in a single replacement reaction, creating aluminum chloride and copper. The aluminum, which was in parallel with the LEDs, breaks and electricity cannot travel through it. So, the electricity in the circuit now goes through the LEDs on the tree instead, lighting them.
Reaction 5: We used copper nitrate and ammonium sulfate to create ammonium nitrate and copper sulfate. This was a double displacement reaction and created a solid, which is called a precipitate.
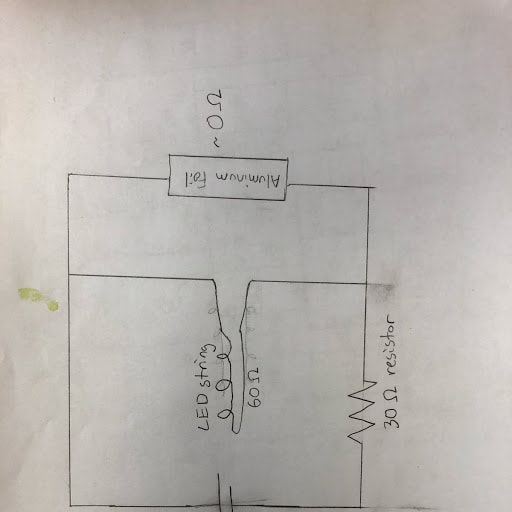
This is a picture of our circuit. We have a resister on the LED line so the current goes through the aluminum, which has almost zero resistance. When the aluminum is broken, the current goes through the LED.
KEY TERMS AND FORMULAS:
Combustion reaction: Hydrocarbon + O2 to CO2+H2O
Single replacement reaction: A+BX to AX+B
Double replacement reaction: AX+BY to AY+BX
Synthesis reaction: A+X to AX
Decomposition: AX to A+X
CHEMICALS AND IONS:
Ammonium Sulfate: NH4(SO4)
Copper Nitrate: Cu(NO3)2
Copper Chloride: CuCl
Aluminum: AL
REFLECTION:
This project in my opinion was very productive and fun. The project helped me learn reactions a lot better. Using the science surrounding us is a much better way to learn than straight out of a textbook, which really helped. My group worked very smoothly and productively throughout the entirety of the project. With the amount of time we had, we put together a very cool board game! Two peaks were our collaboration and work ethic. We were always on time and on the same page when it came to organization and each others ideas. One pit is leadership. I usually didn't take charge during the project. This project overall was very interesting and it was a mix of fun games and chemistry mixed together. It was also very fun to see all of the other group's projects, because there were so many choices on how to do this project. The building aspect was my favorite because i enjoyed it so much in earlier years.
Combustion reaction: Hydrocarbon + O2 to CO2+H2O
Single replacement reaction: A+BX to AX+B
Double replacement reaction: AX+BY to AY+BX
Synthesis reaction: A+X to AX
Decomposition: AX to A+X
CHEMICALS AND IONS:
Ammonium Sulfate: NH4(SO4)
Copper Nitrate: Cu(NO3)2
Copper Chloride: CuCl
Aluminum: AL
REFLECTION:
This project in my opinion was very productive and fun. The project helped me learn reactions a lot better. Using the science surrounding us is a much better way to learn than straight out of a textbook, which really helped. My group worked very smoothly and productively throughout the entirety of the project. With the amount of time we had, we put together a very cool board game! Two peaks were our collaboration and work ethic. We were always on time and on the same page when it came to organization and each others ideas. One pit is leadership. I usually didn't take charge during the project. This project overall was very interesting and it was a mix of fun games and chemistry mixed together. It was also very fun to see all of the other group's projects, because there were so many choices on how to do this project. The building aspect was my favorite because i enjoyed it so much in earlier years.